Section 15: Redox Reactions

OIL RIG, Oxidation States, Reducing Agents, Oxidizing Agents, Combination, Decomposition, Combustion, Single Displacement, Hydrogen Displacement, Metal Displacement, Activity Series, Halogen Displacement, Disproportionation
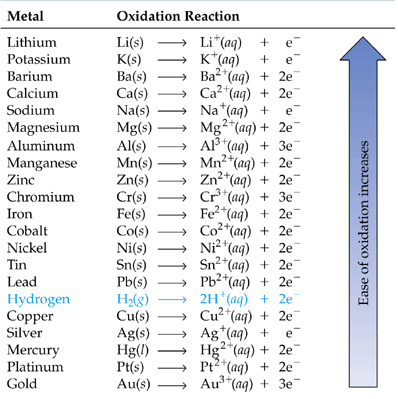
Redox reactions describe what happens when atoms gain or lose electrons. The first step in understanding these types of reactions is to become familiar with the oxidation state numbering system. It is a man made system that helps us understand where are electrons go and where they are coming from. Each of the listed reactions above has different aspects. A combination reaction is when two or more things come together and form one. Decomposition occurs when one compound breaks down into two or more things. Combustion reactions produce flames, it is merely a reaction between oxygen and carbon based compound. Single displacement reactions are essentially the types of reactions that occur in batteries. Electrons are lost by the anode (negative end) and move toward the cathode (positive end) through a wire.
Redox reactions describe what happens when atoms gain or lose electrons. The first step in understanding these types of reactions is to become familiar with the oxidation state numbering system. It is a man made system that helps us understand where are electrons go and where they are coming from. Each of the listed reactions above has different aspects. A combination reaction is when two or more things come together and form one. Decomposition occurs when one compound breaks down into two or more things. Combustion reactions produce flames, it is merely a reaction between oxygen and carbon based compound. Single displacement reactions are essentially the types of reactions that occur in batteries. Electrons are lost by the anode (negative end) and move toward the cathode (positive end) through a wire.
|
Practice
|
|
|
|