Section 17: Liquids & Solids
Section 17A: Intermolecular Forces
Condensed States, Dipole-Dipole, Dispersion Forces, Hydrogen Bonding, Surface Tension, Viscosity, Capillary Action
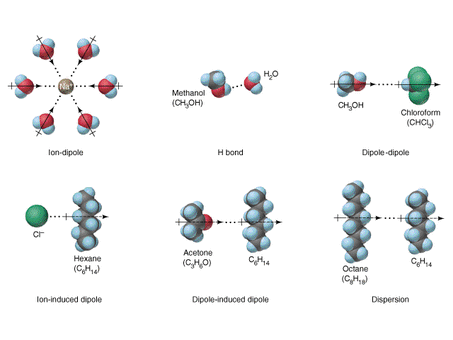
Why is water a liquid at room temperature but oxygen is not? Remember how particle distance plays a role in states of matter of compound. Why is it that at room temperature water molecules are so much closer, yet oxygen particles stay so far apart? The answer of course has to do with intermolecular forces, or the attractive forces that exist between molecules. It should be noted, and emphatically so, intermolecular forces are NOT bonds!! Bonds are classified as the forces that hold two atoms together, whereas IMFs hold molecules close together. These interactions can be measured directly. The boiling point of a substance is a good indication of how strongly molecules are held together for that substance.
Why is water a liquid at room temperature but oxygen is not? Remember how particle distance plays a role in states of matter of compound. Why is it that at room temperature water molecules are so much closer, yet oxygen particles stay so far apart? The answer of course has to do with intermolecular forces, or the attractive forces that exist between molecules. It should be noted, and emphatically so, intermolecular forces are NOT bonds!! Bonds are classified as the forces that hold two atoms together, whereas IMFs hold molecules close together. These interactions can be measured directly. The boiling point of a substance is a good indication of how strongly molecules are held together for that substance.
ForceDipole-Dipole Interactions
Dispersion Forces (London Forces) Hydrogen Bonds Ionic Bonding |
DefinitionThe force that exists between two polar molecules.
The major force between non polar molecules. Exist due to instantaneous dipoles that occur as electrons move around a particle. This is a special type of dipole-dipole interaction. A hydrogen atom must be directly bonded to fluorine, oxygen, or nitrogen, making H-bonding FON! :) Occurs only in ionic compounds. Each ion is attracted to all the other oppositely charged ions around itself. The strongest intermolecular force. |
ExamplesHydrochloric Acid, Formaldehyde
Butane Octane Nitrogen Water Ethanol Hydrofluoric Acid Sodium Chloride Magnesium Oxide Potassium Carbonate |
Section 17B: Solids

Ionic, Covalent Network, Molecular, Metallic
There are four different types of solids that can be spoken of. Each has different characteristics and each substance (when in the solid form) falls into one of these categories. The properties of the solid determine which category the solid is placed.
There are four different types of solids that can be spoken of. Each has different characteristics and each substance (when in the solid form) falls into one of these categories. The properties of the solid determine which category the solid is placed.
Solid TypeIonic
Covalent Network Molecular Metallic |
PropertyBrittle, High MP, Poor Conductors, Good Conductors when dissolved in water, Ionic Bonds
Hard, High MP, Poor Conductors, Atoms connected through network of covalent bonds. Low MP, Poor Conductors, IMFs hold molecules close together Soft - Hard, generally High MP, Good Conductors, Electron-Sea Model of Bonding |
ExamplesSodium Chloride, Magnesium Sulfate, Lithium Nitrate
Diamond, Graphite, Quartz Water, Wax Iron, Sodium, Aluminum, Gold |
|
Resources
|
Practice
|
Section 17C: Phase Changes
Vocabulary, Phase Diagrams, Supercritical Fluids, Vapor Pressure, Change in Enthalpy, Exothermic, Endothermic, Heat Curve
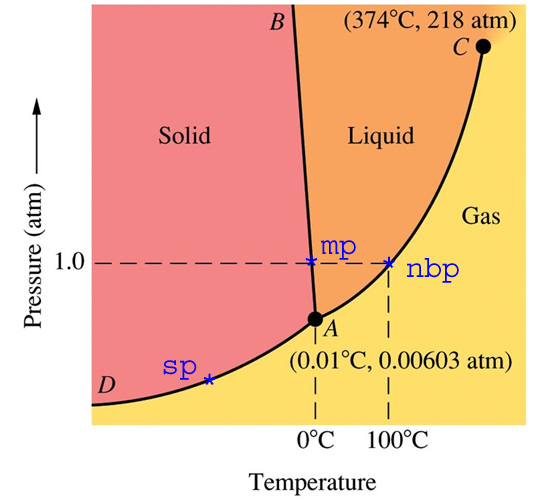
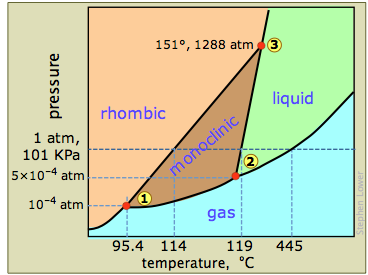
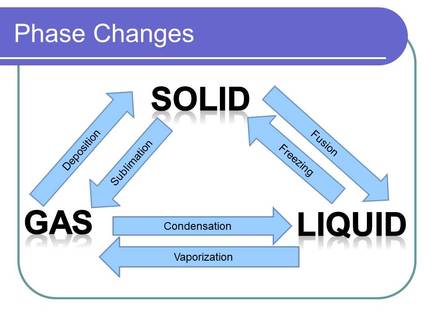
It is known what forces hold compounds together, but what happens when these forces are overcome. This can be visualized in several different ways. The most common is a phase diagram. This is a graph that provides information on the state of matter of a compound at a given temperature and pressure. Below are three phase diagrams. Each shows qualities of the compound that it represents. With phase changes comes different vocabulary. Each phase change has a unique name, that name describes what is happening to a compound on a molecular level. The two words that are important to discussions in chemistry are exothermic and endothermic. Each describes how energy flows in a process. Three of the phases changes are exothermic and three are endothermic.
It is known what forces hold compounds together, but what happens when these forces are overcome. This can be visualized in several different ways. The most common is a phase diagram. This is a graph that provides information on the state of matter of a compound at a given temperature and pressure. Below are three phase diagrams. Each shows qualities of the compound that it represents. With phase changes comes different vocabulary. Each phase change has a unique name, that name describes what is happening to a compound on a molecular level. The two words that are important to discussions in chemistry are exothermic and endothermic. Each describes how energy flows in a process. Three of the phases changes are exothermic and three are endothermic.