Section 18: Solution Properties
Section 18A: Solutions
Solution Vocabulary, Solution Process, Solubility, Mass %, Mole Fraction, Molarity, Dilution, Molality, Gas Solubility, Solubility Rules, Solubility vs Temperature
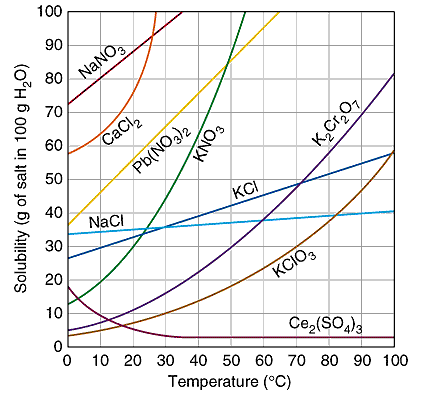
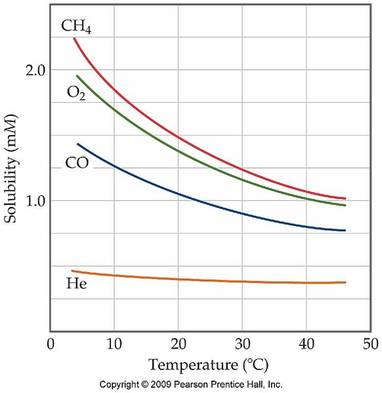
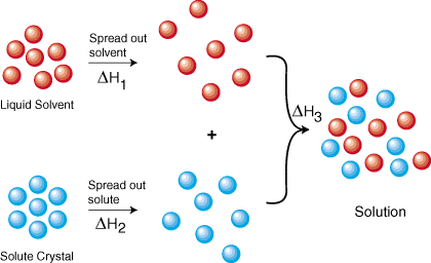
Solutions are the most common state of matter in a lab. Many of the compounds that are found in the stock room are first dissolved in water before they are used in an experiment. There are several parts of the solution process. First the solvent particles must spread out and make room for the solute particles. Secondly the solute particles must break apart and move through the solvent. Finally, the reason that solutions can be made is the solvent and solute particles are attracted to each other, lowering the energy of the overall system. Once a compound has been dissolved, there are several methods that are used to determine the amount of solute that has been dissolved in a given amount of solvent. These methods are listed above, and each is used differently depending on what an individual is trying to do. There are a simple set of rules that are used to predict if a particular compound is soluble in water or if it will remain in the solid phase. These solubility rules will be important, especially for double replacement reactions. The graphs below show the solubility of both solids and gases in water according to temperature.
Solutions are the most common state of matter in a lab. Many of the compounds that are found in the stock room are first dissolved in water before they are used in an experiment. There are several parts of the solution process. First the solvent particles must spread out and make room for the solute particles. Secondly the solute particles must break apart and move through the solvent. Finally, the reason that solutions can be made is the solvent and solute particles are attracted to each other, lowering the energy of the overall system. Once a compound has been dissolved, there are several methods that are used to determine the amount of solute that has been dissolved in a given amount of solvent. These methods are listed above, and each is used differently depending on what an individual is trying to do. There are a simple set of rules that are used to predict if a particular compound is soluble in water or if it will remain in the solid phase. These solubility rules will be important, especially for double replacement reactions. The graphs below show the solubility of both solids and gases in water according to temperature.
|
BrightStorm
Practice
|
Resources
|
Section 18B: Colligative Properties
Vapor Pressure Lowering, Boiling Point Elevation, Melting Point Depression, Osmotic Pressure, Colloids
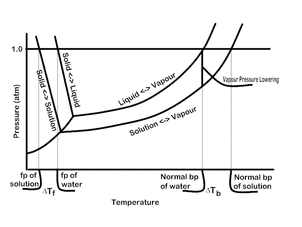
Why is salt put on the roads during the winter months? Why does salt have the ability to melt the ice on the roads? What salt is used when this operation is done? There are several properties of solutions that have been harnessed. Not only can salt melt ice, but is can increase the amount of energy needed to boil a liquid as well as increase osmotic pressure (a topic more appropriate for biology). These properties are dictates by the amount of particles that are dissolved in water. Why is salt used and not sugar? The fact that salts break into their ions means that when salt is dissolved in water, there are more particles produced than if a substance that does not break down is placed in water.
Why is salt put on the roads during the winter months? Why does salt have the ability to melt the ice on the roads? What salt is used when this operation is done? There are several properties of solutions that have been harnessed. Not only can salt melt ice, but is can increase the amount of energy needed to boil a liquid as well as increase osmotic pressure (a topic more appropriate for biology). These properties are dictates by the amount of particles that are dissolved in water. Why is salt used and not sugar? The fact that salts break into their ions means that when salt is dissolved in water, there are more particles produced than if a substance that does not break down is placed in water.
|
BrightStorm
Practice
|
Resources
|