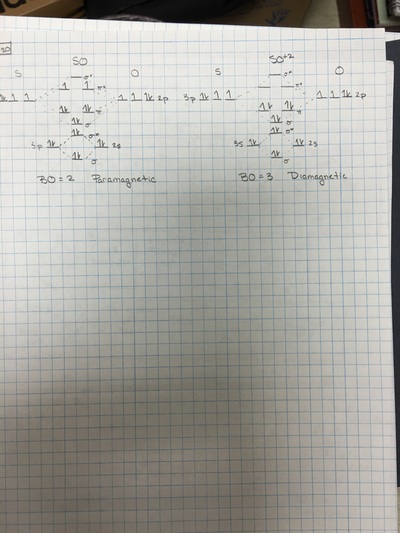Section 10: Molecular Geometry
VSEPR. 3D Drawing, Polar Molecules
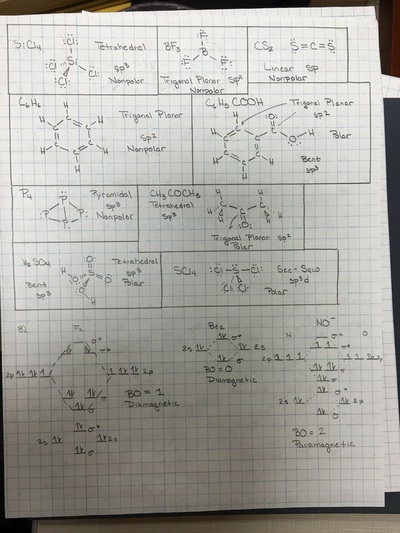
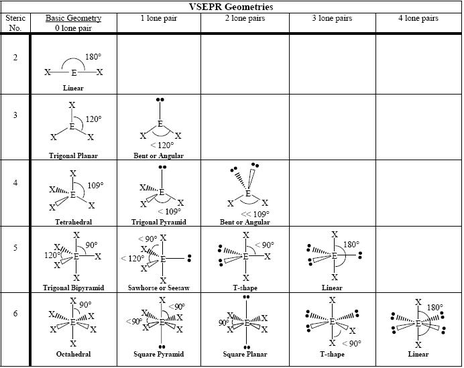
Many of the properties of molecules are derived from the arrangement of atoms in three dimensional space. Many biological reactions are restricted to molecules of specific shape. Valence Shell Electron Pair Repulsion is the accepted theory that describes molecular shape. The geometry of a molecule will determine if a molecule is polar or not. Polarity plays a major role in a molecule's ability to dissolve in water. The ability to dissolve a molecule is water is an important aspect that many drug companies deal with. If a company is trying to deliver a molecule to a specific area of the body, it must be determined if a molecule will dissolve in water, thus have the ability to travel in the blood. If the molecule cannot be dissolved, then does it have to be injected into a specific area or is there another delivery method?
Many of the properties of molecules are derived from the arrangement of atoms in three dimensional space. Many biological reactions are restricted to molecules of specific shape. Valence Shell Electron Pair Repulsion is the accepted theory that describes molecular shape. The geometry of a molecule will determine if a molecule is polar or not. Polarity plays a major role in a molecule's ability to dissolve in water. The ability to dissolve a molecule is water is an important aspect that many drug companies deal with. If a company is trying to deliver a molecule to a specific area of the body, it must be determined if a molecule will dissolve in water, thus have the ability to travel in the blood. If the molecule cannot be dissolved, then does it have to be injected into a specific area or is there another delivery method?
Section 10: Orbital Overlap
Sigma and Pi Bonds, Hybridization, Effect on Geometry
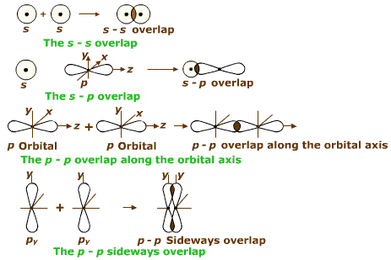
This bonding theory is what is used to describe the different geometries that molecules can take. If a covalent bond is formed when two atoms share electrons, why can carbon make four bonds? The electron configuration for carbon indicates that there are two paired electrons and two unpaired electrons.
This bonding theory is what is used to describe the different geometries that molecules can take. If a covalent bond is formed when two atoms share electrons, why can carbon make four bonds? The electron configuration for carbon indicates that there are two paired electrons and two unpaired electrons.
Section 11B: Molecular Orbital Theory
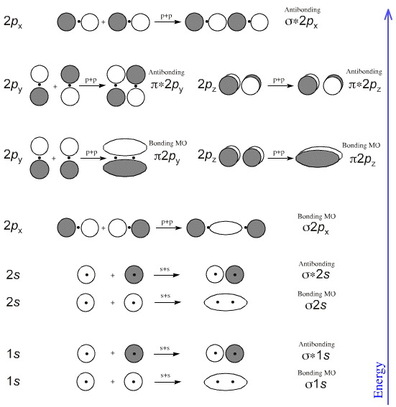
MO Orbitals, MO Diagrams, Bond Order, Para/Dia-magnetic
This theory describes different characteristics of diatomic molecules. Although it is possible to describe larger molecules with this theory, it becomes exceedingly difficult. One of the properties of diatomic molecules that MO Theory describes well is magnetism. Many compounds that, through Orbital Overlap Theory appear to be diamagnetic, in actuality are attracted to magnets. The video below shows what happens when liquid oxygen is poured between two magnetic plates.
This theory describes different characteristics of diatomic molecules. Although it is possible to describe larger molecules with this theory, it becomes exceedingly difficult. One of the properties of diatomic molecules that MO Theory describes well is magnetism. Many compounds that, through Orbital Overlap Theory appear to be diamagnetic, in actuality are attracted to magnets. The video below shows what happens when liquid oxygen is poured between two magnetic plates.